Bohr Model Worksheet 1 What two sub atomic particles are located in the nucleus of the atom 2 What is the difference between the atomic number the mass number of an element 3 Where is
Q1 What part of the periodic table tells you the number of protons found in the nucleus of the atom of that element Q2 By using the periodic table how did you determine how many neutrons are in the most common isotope of an element Q3 How many electrons are needed to fill the first energy level The second energy level Bohr model Other contents physics Share Print Worksheet Finish Liveworksheets transforms your traditional printable worksheets into self correcting interactive exercises that the students can do online and send to the teacher
Bohr Model Worksheet

Bohr Model Worksheet
https://i.pinimg.com/originals/e5/d1/7b/e5d17b3a0cdf6eb055c8f7eb456623ba.jpg

What Is The Bohr Model Worksheet And Answers Style Worksheets
https://i.pinimg.com/originals/2a/93/66/2a9366ec7cea3a379c6812ba54d97007.jpg

Bohr Model Diagram Worksheets
https://i.pinimg.com/originals/6a/ae/aa/6aaeaaf552f84f12bb0131998317fd36.jpg
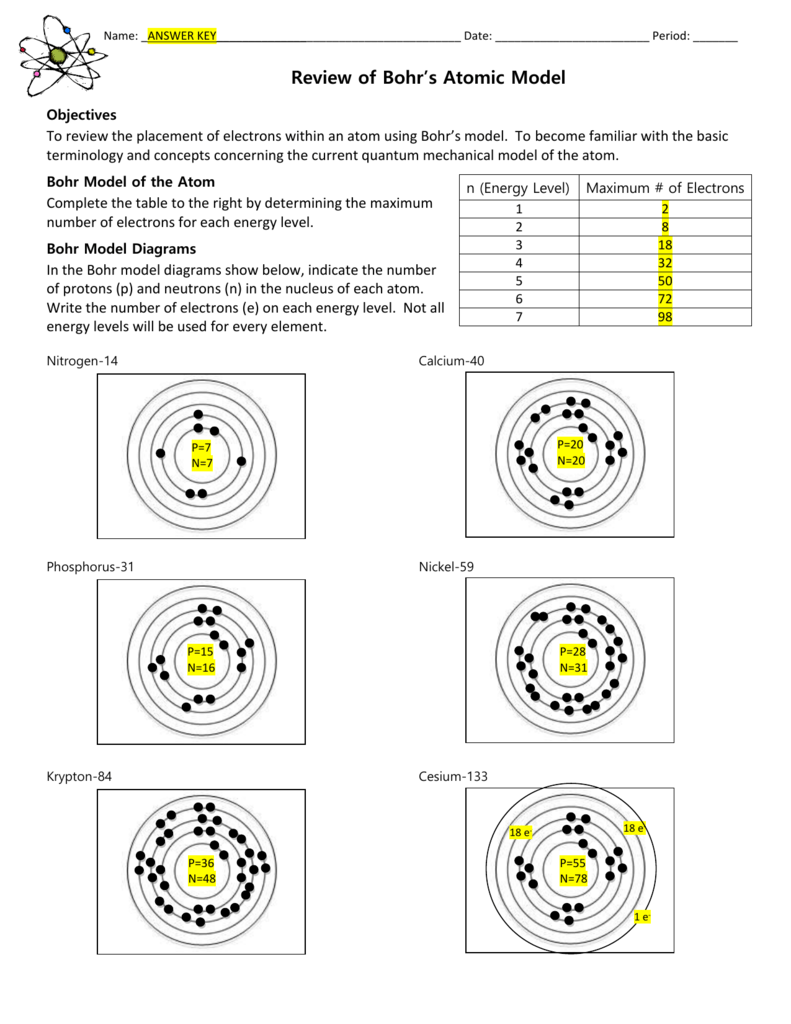
PDF This worksheet is a great way to check knowledge of basic vocabulary terms as well as check for understanding with Bohr Models There is one page of vocabulary terms and two pages of Bohr Model Identification There are 12 different Bohr Models represented in this worksheet as well as 9 terms The Bohr model assumes that the electrons move in circular orbits that have quantized energies angular momentum and radii that are specified by a single quantum number n 1 2 3 but this quantization is an ad hoc assumption made by Bohr to incorporate quantization into an essentially classical mechanics description of the atom Bohr
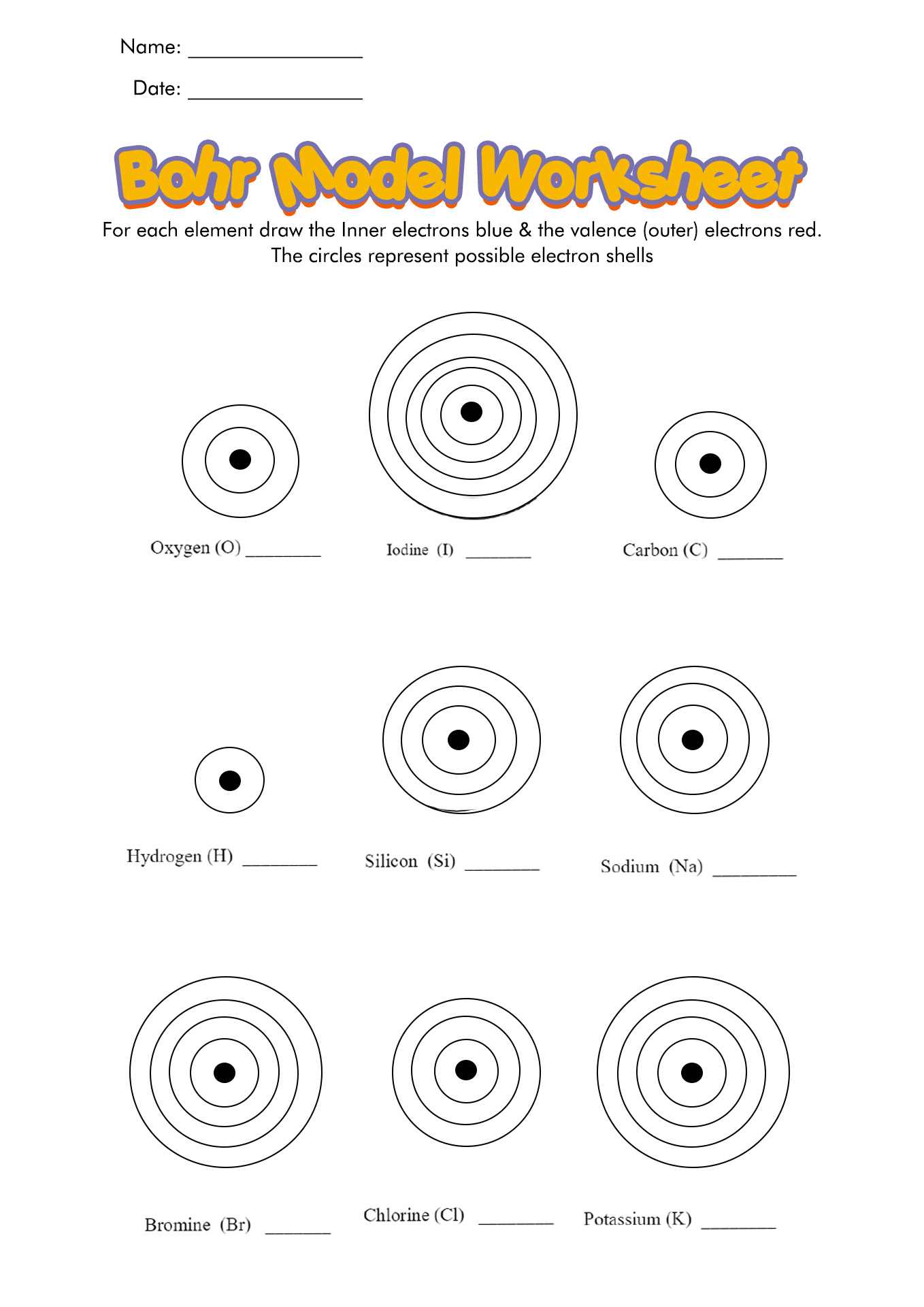
Bohr Model Practice For each element write the total number of electrons on the line Then color the correct number of electrons for each orbit Remember fill the orbit closest to the nucleus first but never exceed the number each orbit can hold Check the Periodic Table to find out how many electrons each element actually has Bohr Model Worksheet Packet 4 Versions of 3 Practice Worksheets Differentiated Skill Levels ideal for your students Can be used for Scaffolded LessonsVery Easy Bohr Model Practice Worksheets 1A 1B and 1C 6 Exercises each 18 exercises total Students given Periodic Table of Element Squares Atomic Number Atomic Mass
More picture related to Bohr Model Worksheet
18 Best Images Of Bohr Diagram Worksheet Bohr Model Worksheet Answers
http://www.worksheeto.com/postpic/2013/09/bohr-model-worksheet-answers_211771.png

Bohr Model Diagrams Worksheet Losangeles sincity
https://i.pinimg.com/originals/0e/c0/b4/0ec0b4845f6e31896e416038b5ae7357.jpg
18 Best Images Of Bohr Diagram Worksheet Bohr Model Worksheet Answers
http://www.worksheeto.com/postpic/2013/09/atomic-structure-bohr-model-worksheet_211787.png
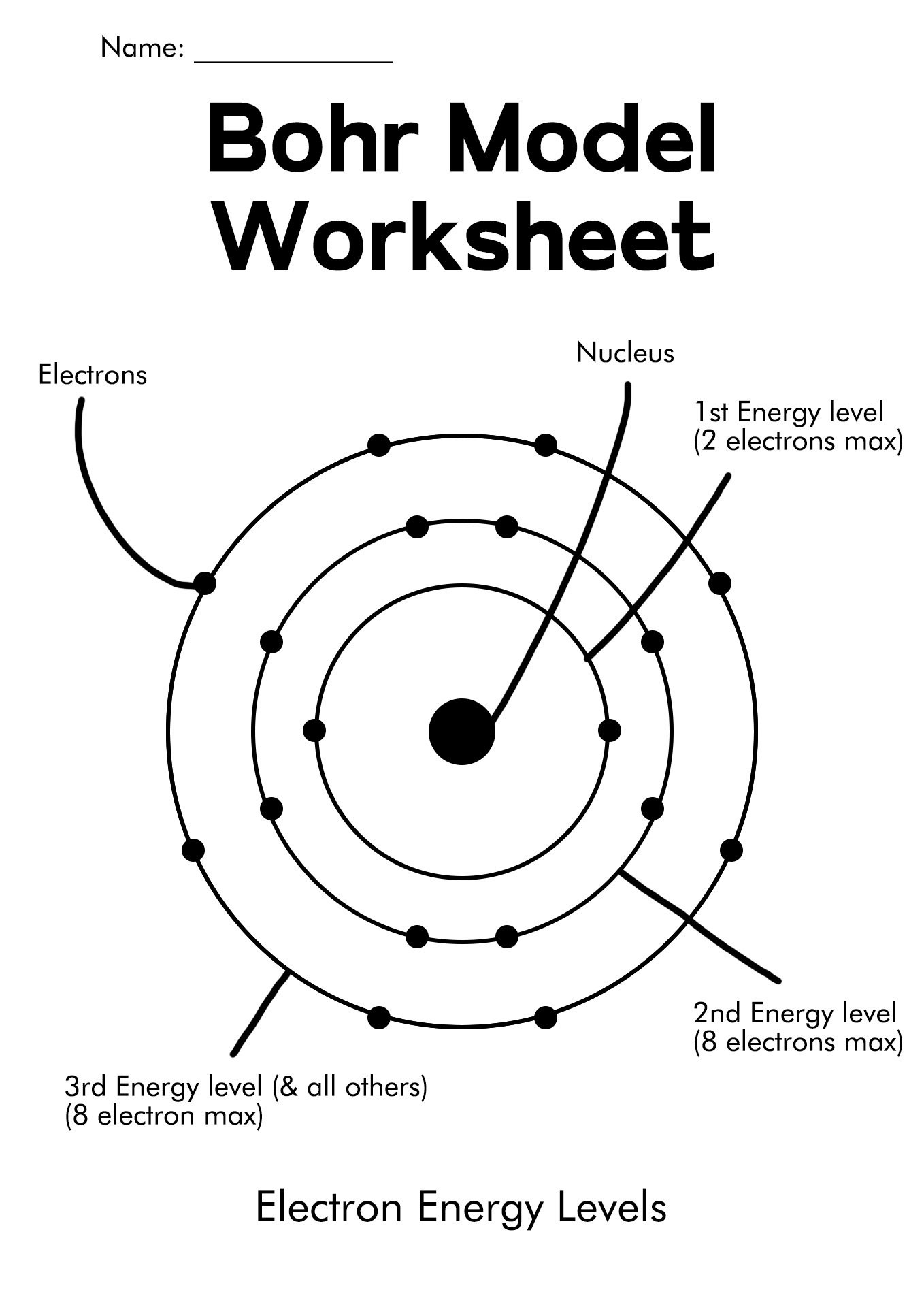
The Bohr s model of the atom Venkata Member for 3 years Age 9 13 Level GRADE 9 Language English en ID 480553 07 11 2020 Country code AE Country United Arab Emirates School subject Science 1061951 Main content Atomic and emission spectra 1191345 Follow the instruction and answer the questions Share Print Bohr Model Diagrams Draw a nucleus with the element symbol inside Write the number of Protons and Neutrons the element has inside the nucleus Put a by the P and a little o by the N Note Round mass to nearest 1 when figuring neutrons hold a
This chemistry worksheet is perfect for more practice on the atomic chart Bohr s Model Can be use digitally drag and drop or in person Practice for the first 3 periods of the periodic table The Bohr model worked beautifully for explaining the hydrogen atom and other single electron systems such as He text He He start text H e end text start superscript plus end superscript Unfortunately it did not do as well when applied to the spectra of more complex atoms
Bohr Model Worksheet Answers
https://s3.studylib.net/store/data/008594319_1-04b3e75ec02db0292a787df2a035aa12.png

Worksheet Bohr Models
https://s3.studylib.net/store/data/008197589_1-c356d1d576850a8c5bf065a08755c2c9.png
Bohr Model Worksheet - Bohr model ID 1771521 Language English School subject Physics Grade level 3 Age 15 16 Main content Atomic Other contents physics Add to my workbooks 0