Calculating Particles In The Nucleus Worksheet Answer Key Part l Label the parts of this atom nucleus protons electrons neutrons Part 2 Answer these 1 The subatomic particle with no electrical charge is the 2 The subatomic particle with a positive charge is the 3 The subatomic particle with a negative charge is the 4 There are the same number of these two particles in an atom 5
Strong Nuclear Forces Teacher Answer Key 1 What three particles make up an atom and what are the electrical charges of the three particles Protons neutrons and electrons Protons have a positive charge Neutrons have a neutral charge 0 Electrons have a negative charge 2 Showing 8 worksheets for Calculating Particals In The Nucleus Worksheets are Km 654e 20150109102424 Nuclear physics work answers Protons neutro Calculating Particals In The Nucleus Worksheets K12 Workbook
Calculating Particles In The Nucleus Worksheet Answer Key
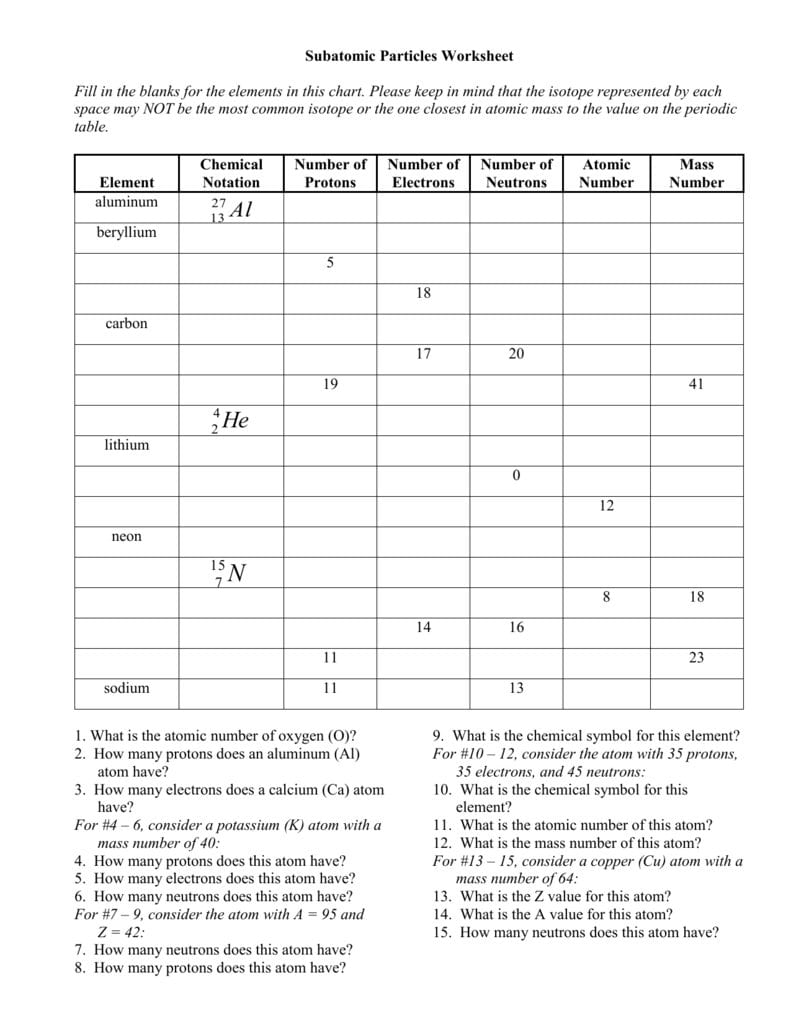
Calculating Particles In The Nucleus Worksheet Answer Key
https://db-excel.com/wp-content/uploads/2019/09/subatomic-particles-worksheet-2.jpg

48 Calculating Particles In The Nucleus Answer Key CahraAllysa
https://s3.studylib.net/store/data/005887600_1-11e56d7c4c198783e62d7f2f192dff90.png

Atomic Structure Chart Worksheet Answers Cpo Chapter 14 Physical
https://i1.wp.com/online.anyflip.com/vybm/mmvr/files/mobile/3.jpg
Atomic Basics Answer Key Part A Atomic Structure 1 Draw five protons in the nucleus of the atom Label them with their charge 2 Draw six neutrons in the nucleus of the atom 3 Draw two electrons in the first energy level and label them with their charge 4 Draw three electrons in the second energy level and label them with their charge 5 Expert Answer Calculating partieles in the nucleus Name Atomic mass total number of particles in the nucleus Atomic mass atomie number the number of neutrons Complete the table Element Atomic Atomic Mass 12 Number f Number of Number of Neutrons Electrons symbol Protons Cu Pb Xe Tc 29 82 35 207 131 82 43 12 77 115 43 12 24 3 27
In the nucleus repel push away from each other more So are required to make the nucleus stable Neutrons are though to be needed to hold all the protons together in the nucleus The bigger the nucleus the True of False All matter is made of small particles called atoms Atoms of one elements are different form the atoms of other elements Calculating Particles In The Nucleus Displaying top 8 worksheets found for this concept Some of the worksheets for this concept are Chapter 4 lesson 1 protons neutrons and electrons Chapter 2 the atomic nucleus Calculating particles in the nucleus work answers Chapter 2 atoms molecules and ions Atoms family work Atom work Answers
More picture related to Calculating Particles In The Nucleus Worksheet Answer Key
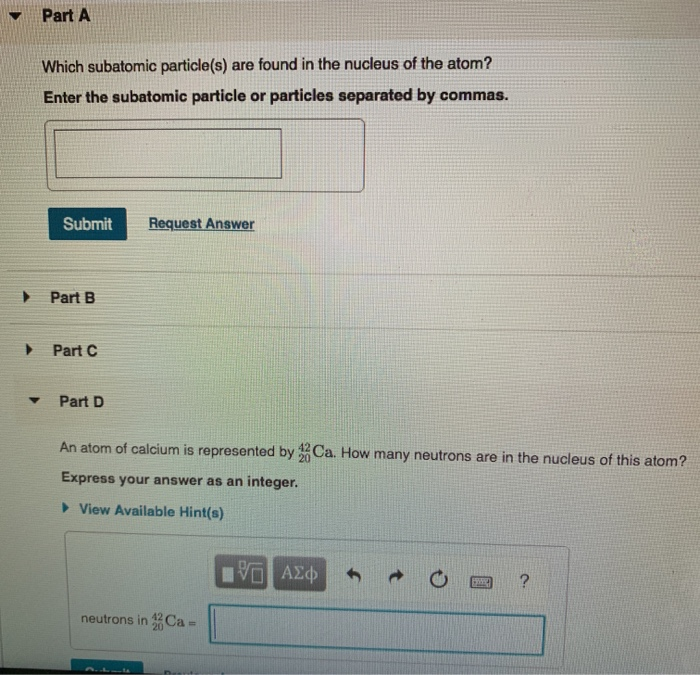
48 Calculating Particles In The Nucleus Answer Key CahraAllysa
https://media.cheggcdn.com/study/a8e/a8efeef6-27db-4122-8c2f-bc6573b241bc/image.png

48 Calculating Particles In The Nucleus Answer Key CahraAllysa
https://s3.studylib.net/store/data/008950387_1-94f1fe17737aacaecfd86bf832f453e9-768x994.png

48 Calculating Particles In The Nucleus Answer Key CahraAllysa
https://images.twinkl.co.uk/tw1n/image/private/t_630_eco/image_repo/9b/85/t3-sc-704-egg-experiments-secondary-easter-resources-preview_ver_5.jpg
Nuclear Physics Worksheet Answers The nucleus plural nuclei is the core of the atom and is composed of particles called nucleons of which there are two types protons positively charged the number of protons in a nucleus is called the Atomic Number symbol Z and identifies which element corresponds to that particular nucleus Displaying 8 worksheets for Calculating Particals In The Nucleus Worksheets are Km 654e 20150109102424 Nuclear physics work answers Protons neu
15 How many neutrons are in the nucleus of an atom that has an atomic mass of 36 and an atomic number of 25 Bohr Model Drawing 25 points Draw a Bohr model of an oxygen atom in the space below Be sure to place the electrons in the correct orbitals and to fill out the key for the subatomic particles Key Protons Neutrons Electrons Nucleus Unit 4 The Nucleus key Regents Chemistry 14 Z15 Mr Murdoch Page 9 of 55 Website upload 2014 2 Particles outside the Nucleus i Electrons Electrons have a mass of 1 1836th amu 9 11 x 10 28 grams and a nuclear charge of 1 Electrons are found orbiting the nucleus in energy levels Atoms gain lose or share electrons when they form

48 Calculating Particles In The Nucleus Answer Key CahraAllysa
https://s3.studylib.net/store/data/008110349_1-3972f772d67c1949a491b0b2a0203a7e-768x994.png
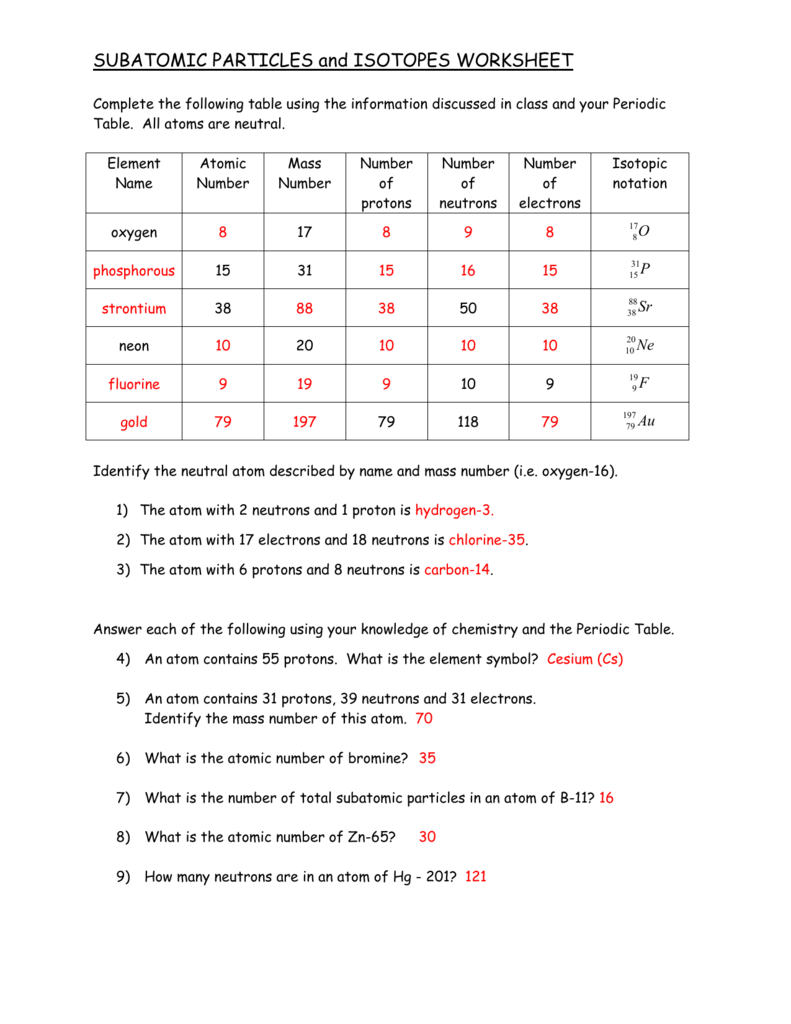
Subatomic Particle Worksheet Answers Educational Worksheet
https://s3.studylib.net/store/data/008278762_1-a13b9cb8a43d70165cc82735cfe5e185.png
Calculating Particles In The Nucleus Worksheet Answer Key - Nuclear Physics Worksheet The nucleus plural nuclei is the core of the atom and is composed of particles called nucleons of which there are two types protons positively charged the number of protons in a nucleus is called the Atomic Number symbol Z and identifies which element corresponds to that particular nucleus